Overarching Cancer Research and Development Ambitions Strategy 2021-31
Overarching Cancer Research and Development Ambitions Strategy 2021 -31
Executive summary
Velindre has a key role to play in the cancer research network in South East Wales (SEW). It provides an important link between the 3 University Health Boards in the region (Cardiff & Vale, Aneurin Bevan and Cwm Taf Morgannwg UHBs) for collaborative clinical cancer research, offering opportunities for patients to access clinical trials and a range of other research studies, either at Velindre Cancer Centre (VCC) itself or in outreach facilities at the UHBs. Velindre is also in a prime position to provide the crucial connection between laboratory cancer researchers and patients, enabling research to ‘bridge the translational gap’ and bring new discoveries from the laboratory to the clinic for patient benefit.
Our vision is to work with patients and partners to design and deliver excellent research that improves the survival and enhances the lives of patients and their families.
Velindre is a patient focused organisation which performs consistently well in patient satisfaction questionnaires. As an organisation however we have not maximised opportunities to offer every patient access to research and to inter-link our research to maximise the learning opportunities from every patients’ data and experience. Putting patients first and at the centre of everything we do and maximising every patient contribution will be a future strategic research priority. Velindre has a strong track record of leading national and international practice-changing cancer Clinical Trials, led by Velindre researchers and Sponsored by the organisation. We will build on this to develop the next generation of multi-disciplinary researchers in order to advance new treatments, interventions and care. The ability to seamlessly translate discovery research from the laboratory to the clinic for patient benefit and to take patient samples (or images/technology/knowledge) back to the laboratory to generate new knowledge has not previously been fully exploited. Driving translational research by enabling closer partnerships between academic cancer researchers in the University and clinical cancer researchers in the NHS is a future strategic priority. To optimise our ambitions it will essential to ensure research is embedded within the organisational culture and structures.
The Nuffield advice (published 1 Dec 2020) highlighted the importance of research and development (R&D) for delivering patient centred, future-proofed, high quality cancer care for people in SEW. The new Velindre Cancer Centre must be designed to enable R&D activity and make research available to all patients attending the centre. Nuffield recommended the development of “a strong research hub at UHW” to deliver cancer Early Phase Clinical Trials and Advanced Therapies, co-located with HDU/ITU. A tripartite Cardiff Cancer Research Hub will enable this, “collaboration with haemato-oncology research” and “opportunities for closer working with the University”. Nuffield also recommended Velindre@ Research Facilities in all UHBs to be “viewed as a key part of the research delivery network and supported accordingly” in view of their “access to large patient numbers and support from ITU and other specialities”. The Velindre NHS Trust Executive support the Nuffield Trust recommendations which will enable the vision and ambitions laid out in this paper to be delivered, enhancing research opportunities for cancer patients across SEW. The network model will require new infrastructure, regional collaboration between the Trust and UHB partners and a refreshed strategic research partnership between the Trust and Cardiff University which will, if realised, accelerate new discoveries into the clinic and build a critical mass of cancer research activity in SEW that will generate inward investment and growth and could represent a turning point for cancer research in Wales.
1. Summary
1A. Our Vision:
Our vision is to work with patients and partners to design and deliver excellent research that improves the survival and enhances the lives of cancer patients and their families in Wales and beyond.
Our mission is to become a leader in cancer research nationally and internationally, transforming the culture of our organisation into one where every patient, family and staff member who wants to engage with research has the opportunity to do so.
To enable this, we will work with our NHS and academic partners, with a shared strategic focus and collaborative ethos.
1B. Our Aims:
-
- Enhance patient experience and care
- Improve patient outcomes and reduce variation
- Accelerate the implementation of new discoveries into the clinic
- Demonstrate the impact of our research on patients and the NHS
- Build research capacity and capability at Velindre and across South East Wales.
1C. Our Research Themes:
Our research will be divided into 4 interconnected strategic themes:
-
- Putting patients first and at the centre of everything we do
- Advancing new treatments, interventions and care
- Driving translational research through connecting the laboratory and clinic
- Embedding research and innovation within the organisational culture.
Underpinning the work in each theme will be cross-cutting departmental research strategies. High quality research will be facilitated by a governance and enabling infrastructure and delivered by an agile research support workforce based at Velindre and across South East Wales.
1D. This Document:
This document sets out our overarching ambitions for cancer research over the next 10yrs, informed by:
-
- The Velindre Futures Task and Finish Group of multidisciplinary Velindre and Cardiff University cancer researchers and Patient and Public Representatives (see Appendix 1 for group membership and authorship of this document).
- The Nuffield Trust advice on the proposed model for non-surgical tertiary oncology services in South East Wales.
- Welsh and UK key policies and strategies that impact on research and current strategies within Velindre (see Appendix 2).
The ambitions for Welsh Blood Service research, whilst vitally important for the Trust, are not included in this document and will need to be developed separately.
The next step for cancer research will be to develop a detailed implementation plan, in partnership with our University colleagues and University Health Board (UHB) partners across South East Wales. In parallel, we will engage with Charities, Industry and the Welsh Government as key partners in cancer research across the region and beyond.
2. Background
2A. Introduction:
Healthcare research for is vital for both patients and the NHS. It underpins the evidence needed to provide the best care and services for patients. Patients treated in research-active healthcare settings have better outcomes and receive better care, with benefits extending to patients beyond those actively involved in research studies (1, 2). Participating in research empowers patients and >90% have a good experience of taking part in research (3). Patients want opportunities to be involved in trials of new treatments, and the public believes that the NHS should play an important role in supporting research for new treatments (4). Conducting excellent research ensures that clinicians are well versed in research findings and the evidence base for the treatments they give. Research has wider benefits for the NHS (5, 6) - it underpins innovation and service improvements in healthcare, it can improve efficiency and effectiveness and it motivates, attracts and retains staff (7).
The Welsh Government NHS Wales Planning Framework 2020-23 (8) sets out an expectation for research involvement within NHS Wales as follows: “Evidence indicates that research active organisations provide better care and can achieve better patient / public outcomes than those NHS organisations that conduct less research. High quality research can help break the legacy of ill health, develop a prosperous society through collaborative engagement with universities, industry and the third sector. Everyone working in the NHS should regard research as an integral part of their role.” and “organisations will be expected to demonstrate how the workforce are being supported to undertake research and how research and development is informing their planning, financial and decision making.”
Cancer is the leading cause of death in Wales and incidence rates are rising at around 2% a year, as are the overall number of people in Wales living with cancer. Survival rates in Wales are poor compared to other European nations and there are significant regional disparities in incidence, prognosis and cancer outcomes across Wales. The Cancer Delivery Plan for Wales (2015-2020) states that: “Cancer research is critical to improve outcomes for patients and for the health of people in Wales. Excellent research delivers high impact outputs that change future cancer understanding, treatment and care interventions across the whole breadth of the patient pathway, from primary through to palliative care. People in Wales affected by cancer should have equitable access to participate in clinical trials and other well designed studies.”
2B. Context:
Velindre has a key role to play in the cancer research ecosystem in Wales (Figure 1). It is the largest provider of non-surgical oncology treatment in Wales, receiving 6000-7000 new cancer patient referrals per annum combined from Cardiff and Vale, Aneurin Bevan and Cwm Taf Morgannwg University Health Boards (UHBs). Described within Velindre Cancer Centre’s strategy ‘Shaping our Future Together 2026’ is a vision to be an “International leader in research, development, innovation and education.” Velindre University NHS Trusts’ (VUNHST) strategic priority within the Integrated Medium Term Plan (IMTP) 2019-2022: is to “transform the organisation into one synonymous with excellence in research, development and innovation at a scale well-beyond our current offering”.
Excellent research requires partnerships and through its Academic Partnership Board (established in 2020) Velindre has committed to support future collaboration with Academic partners across Wales. A Memorandum of Understanding (MOU) between VUNHST and Cardiff University (CU) describes a shared commitment to develop an ever-closer strategic partnership between both organisations, bringing mutual benefits for education and research. Other key research partners include NHS Wales, Welsh Government via Health and Care Research Wales (HCRW), the Third Sector, the Wales Cancer Research Centre (WCRC), the Wales Cancer Network, (WCN), the Wales Cancer Bank (WCB), Public Health Wales, the Rutherford Centre, Genomics Partnership Wales and the Life Sciences Hub. Commercial partnerships are key in research, in terms of technology, patient experience, pharma trials and innovation.
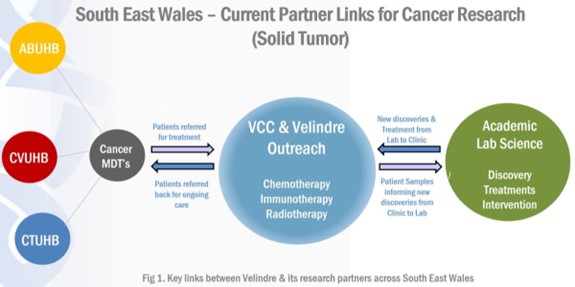
Figure 1: Key links between Velindre and its research partners across South East Wales
Transforming Cancer Services (TCS), the new Radiotherapy Satellite Centre and the proposed new Velindre Cancer Centre (nVCC) funded through Welsh Government capital investment, provide excellent research and innovation opportunities. Such new infrastructure, if paired with investment in the research workforce and a shared and reciprocal commitment between NHS organisations and academic partners to collaborate on a regional approach to cancer research and development (R&D) could provide the necessary critical mass required to drive and deliver a step change in Wales’ future cancer research outputs.
2C. Current research strengths:
Velindre has a legacy of excellence in Clinical Trial leadership and our trials have influenced clinical practice nationally (9) and internationally (10). The current portfolio of clinical trials led and Sponsored by Velindre include PATHOS (11) and PEARL in head and neck cancer, CORINTH in anal cancer, SCOPE 2 in oesophageal cancer, ‘MRI mapping of metabolic function in brain tumours’ and STORM_Glio in brain cancer and iNATT (international Anaplastic Thyroid Cancer) in thyroid cancer (see Appendix 3 and 4A).
The 1st solid cancer Early Phase Clinical Trial Treatment Unit in Wales opened at Velindre in 2013, delivering a portfolio of Phase I-II Clinical Trials to test the safety and efficacy of novel agents, either alone or in combination with radiotherapy, in collaboration with Cardiff’s Experimental Cancer Medicine Centre (ECMC), Wales Cancer Research Centre (WCRC) and Pharma e.g. FAKTION for metastatic breast cancer (12). As well as leading clinical trials, Velindre currently hosts >100 early to late phase studies led by other organisations, enabled by Velindre Principal Investigators (PIs) (see Appendix 4C and D).
Radiotherapy research at Velindre involves the multi-disciplinary radiotherapy workforce. Medical physics researchers have developed novel tools (e.g. EdgeVCC [13]) to automate radiotherapy planning and our research partners at CU School of Engineering are experts in computing and data analysis. Velindre is one of 4 UK centres which form the National Radiotherapy Trials Quality Assurance (RTTQA) group, leading national RTTQA programmes for gastro-intestinal and head and neck cancers. Together with research partners at CU’s Brain Research Imaging Centre (CUBRIC) and Wales’ PET Imaging Centre (PETIC) our researchers have led studies to understand the effects of brain radiotherapy on neurocognitive function and to adapt radiotherapy mid-treatment based on tumour response in patients with head and neck cancer. Recently, partnerships have been developed with cancer researchers in the immunology, virology and biosciences laboratories at CU, through jointly funded PhD studentships and Clinical Research Fellowships.
For Patient focused Palliative and Supportive Care Research Velindre has close links with the Marie Curie Palliative Care Research Centre (MCPRC) one of 2 core funded UK MCPRCs. It has a track record of studies assessing late effects of radiotherapy (EAGLE -Evaluating and Addressing Gastrointestinal Late Effects of pelvic radiotherapy), embedding qualitative studies within Phase III oncology trials to better understand patient experience and treatment trade-offs (e.g. ROCS and FOCUS IV trials), developing screening tools (e.g. ALERT-B - Assessing Late Effects of Radiotherapy-Bowel) and core outcome sets (e.g. COBra - Core Outcomes in Brain tumour trials) (see Appendix 3).
With recent advanced and consultant practitioner roles being developed, an important area of multi-professional research is emerging, conducted by nursing (14), Allied Health Professional [physiotherapy, occupational therapy, dietetics, speech and language therapy, radiographer], psychology and pharmacy researchers (referred to in the rest of this document as ‘non-medical’ researchers).
2D. Current challenges:
Over the last decade Velindre has treated an increasing number of cancer patients year on year. Despite our current activity and strengths, most of our patients are not currently offered opportunities to access research studies (see Appendix 4B). Constraints which limit our R&D activity are:
General:
- Personalised medicine (where treatment is tailored to an individual’s cancer) has changed clinical trial design, making trials more focused and reducing the numbers of patients recruited per trial, requiring more trials to be set-up to maximise research opportunities for patients.
- Instability of time-limited research funding and increasing competition for charitable (Third Sector) research funding, further exacerbated by COVID-19.
Local/Regional:
- Lack of a clear strategy and implementation plan for cancer research across the SEW region.
- Lack of joint cancer research strategic focus between Velindre and CU (as well as other academic partners in the region), limiting the translation of research from the laboratory to the clinic and resulting in a lack of well-defined career pathways for ‘interface posts’ between academia and the NHS – including clinical academics, nurse researchers and other multidisciplinary researchers.
- Lack of research capacity, workforce capability, succession planning and of investment in the next generation of researchers.
- An increasing need to conduct certain research activities (including Early Phase Clinical Trials) on an acute hospital site with limited facilities to do so currently.
- A need to conduct other research activities closer to patients’ homes across the SEW region.
- The difficulty of conducting research across organisational boundaries in the SEW region and of suboptimal partnership working.
2E. Future opportunities:
The Nuffield Trust report Dec 2020 highlights the importance of R&D for delivering “patient centred, future-proofed, high quality cancer care” for the population of SEW.
There is now a real opportunity (led by Velindre and its partners) to make a step- change in cancer research in SEW by embracing the Nuffield recommendations to develop:
- A new Velindre Cancer Centre (nVCC) which is designed to enable R&D activity, with the multi-disciplinary workforce capacity and capability to make research widely available to patients attending the centre. Opportunities provided by the proposed Centre for Learning and new fleet of radiotherapy machines and radiotherapy research bunker should be realised.
- A “strong research hub at UHW” which will bring together patients, NHS researchers (from C&VUHB and Velindre) and academic researchers (from CU School of Medicine) in one location. This tripartite hub (potentially called a Cardiff Cancer Research Hub) could provide a focus to join-up cancer research in Cardiff, invigorate the cancer research community and provide facilities for:
- Delivery of Early Phase Trials and Advanced Cellular and non-Cellular Therapies for solid cancer with access to HDU/ITU and specialist services (eg surgery, cardiology, immunology, gastroenterology) to manage the complications of therapy and enabling “collaboration with haemato-oncology research” and Teenage and Young Adult (TYA) services.
- Enabling “closer working with the university”, including clinic, office and meeting space, with direct links to the laboratory, biobank, surgery and interventional radiology.
- An enhanced, integrated, multi-disciplinary Clinical Academic workforce, responsible for designing and delivering cancer research that is led from Wales.
- A multicentre clinical research network across SEW, with Velindre@ Research Facilities in all UHBs and a shared vision and infrastructure to support its activities, enabling equitable access to research for patients across the region. Nuffield recommend that these should be “viewed as a key part of the research delivery network and supported accordingly” in view of their “access to large patient numbers and support from ITU and other specialities”.
Paired with a joint focus and strategy for cancer research in Cardiff and supported by investment from the NHS and Cardiff University, as well as potentially the Third Sector and other funders, the Nuffield advice could, if implemented, represent a turning point for cancer research in Wales.
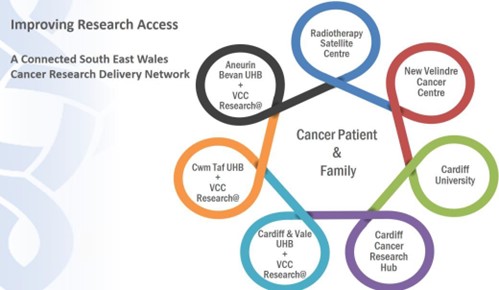
Figure 2: Improving Research Access: A connected South East Wales Cancer Research Delivery Network
3. Realising our vision for research 2021 - 2031
3A. Our Vision and Mission:
Our vision is to work with patients and partners to design and deliver excellent research that improves the survival and enhances the lives of cancer patients and their families in Wales and beyond.
Our mission is to become a leader in cancer research nationally and internationally, transforming the culture of our organisation into one where every patient, family and staff member who wants to engage with research has the opportunity to do so.
3B. Our Aims:
-
- Enhance patient experience and care
- Improve patient outcomes and reduce variation
- Accelerate the implementation of new discoveries into the clinic
- Demonstrate the impact of our research on patients and the NHS
- Build research capacity and capability at Velindre and across South East Wales.

Figure 3: Our research aims
3C. Our Research Themes:
-
- Putting patients first and at the centre of everything we do
- Advancing new treatments, interventions and care
- Driving translational research through connecting the laboratory and clinic
- Embedding research and innovation within the organisational culture
We will increase patient access to research, transform the culture within our organisation into one that can demonstrate that it truly values research and to work with our Academic, UHB and other key partners to produce a step change in our collective cancer research outputs to benefit cancer patients across the SEW region and further afield.

Figure 4: Our research themes
This focus and investment in research will have the following benefits for the organisation:
-
- Ensuring that high quality research results in excellence in care for the wider population VCC serves.
- Generating income which will be re-invested into R&D to build a critical mass of researchers and supporting infrastructure to expand our future research portfolio.
- Enhancing Velindre’s reputation as a centre of excellence committed to improving future cancer patient outcomes.
- Improving staff satisfaction, leading to improved retention and attracting new staff.
Our Aims will be realised by:
-
- Building on our existing strengths, developing our research leaders and multi- disciplinary research workforce.
- Harnessing the potential of new Welsh Government investment in cancer services and implement the Nuffield recommendations to make a step change in our research outputs.
- Working collaboratively with our NHS, Academic and Third Sector partners across SEW and beyond with a shared purpose.
- Focusing our own research efforts on patients undergoing cancer treatment and follow-up but also supporting, where we can, wider research efforts to improve prevention, screening and early diagnosis of cancer.
- Maximising the impact of our research by rapidly implementing new discoveries and innovations into clinical practice for patient benefit.
- Demonstrating the positive impact of our research on patients, the organisation and wider NHS.
4. Our Research Themes in Detail
4A. Theme 1: Putting patients first and at the centre of everything we do
Our research will be driven by the cancer needs of patients. Our service will be designed to enable every patient and family member who wants to engage with research to do so, wherever they live across the region and whatever part of their cancer journey they are on.
What this means:
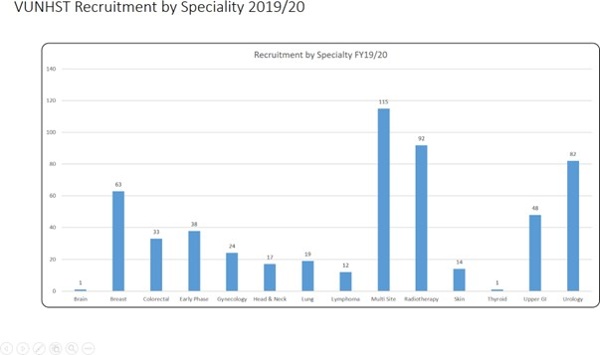
Patients will help set the research agenda. We will increase opportunities for patients and their families to take part in research, so that within 10 years most of our patients are offered research and innovation opportunities at some point in their cancer journey. This represents a significant increase in the percentage of Velindre patients who take part in research. Based on a rolling average of recruitment over the last 5 years and considering the numbers of annual new patient referrals to Velindre, 6-7% of patients referred to Velindre currently take part in research studies (~400 patients per annum, range 304-498 patients, based on 2015-2020 figures – see Appendix 4B). Increasing this to >50% per annum will require a radical change in the organisational culture and regional model for research delivery.
In this theme, we will:
-
- Have a patient and carer focused perspective.
- Improve patient experience
- Reduce inequality & variation in outcomes
- Focus on safe and compassionate care for patients with comorbidity or complex needs and their families.
- Promote a more integrated approach to our research.
-
- What we will do to achieve this:
Have a patient and carer focused perspective by:
-
- Co-producing research with patients and the public, to ensure that our research answers the questions that are important to them and adheres to best practice (15) and UK PPI national standards.
-
- Increasing our focus on Palliative and Supportive Care research, looking for research opportunities throughout the cancer pathway from diagnosis to end of life.
- Extending opportunities to take part in research across the network, through partnership working and Velindre@ research facilities at every UHB, working seamlessly across organisational boundaries to ensure patients can access research nearer their homes.
- Promote the importance of research to patients, the public and staff, through improved communications and engagement to increase participation in research.
Improve patient experience and outcomes by:
-
- Transforming the organisation to enable more patients and families to take part in research.
- Researching the impact of both cancer and treatment side-effects on patient health and wellbeing to understand the trade-offs between disease control and toxicity.
- Understanding the impact of living longer with a cancer on function and wellbeing to help guide future NHS resource use.
- Increasing our observational and qualitative research activity, balancing our portfolio of interventional studies with observational studies.
- Building capacity in the non-medical clinical research workforce in the NHS and across the clinical-academic interface, in collaboration with the MCPRC and the School of Healthcare Sciences at CU
Reduce inequality and variation in outcomes and focus on safe and compassionate care for patients with comorbidity or complex needs and their families by:
-
- Making novel contributions to the evidence base that informs delivery and organisation of cancer care, aiming to make improvements to the lives of patients and their families within 10 years.
- Designing research studies that address inequalities in cancer outcomes, a strategic priority in the Welsh NHS Plan 2020-2023.
- Conducting studies that will focus on the compassionate consultation for shared decision-making and supported self-management to address inequalities in health and well-being outcomes in Wales and beyond.
Promote a more integrated approach to our research by:
-
- Cross-linking research across our themes through integrated and efficient research design to deliver the greatest benefit from every study. As examples, our clinical trial designs will provide patient samples for reverse translation, in turn facilitating new drug discoveries; our Palliative and Supportive Care research on treatment resilience and patient preference integrated with clinical trial design, will inform ongoing trial design and patient approach.
- Considering the clinical, translational and patient focused research questions for every patient, making every patient count more than once to ensure we maximise every patient contribution (see Figure 5 for examples of this approach).
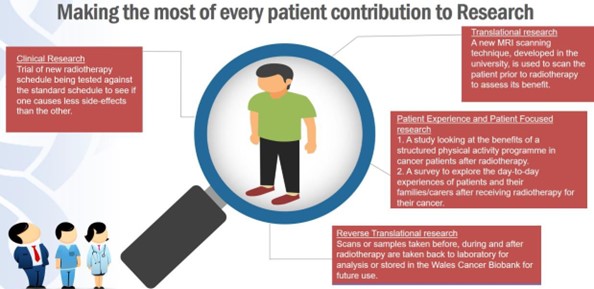
Figure 5: Making every patient count more than once
4B. Theme 2: Advancing New Treatments, Interventions and Care
Our research will be broad and inclusive. Our resources will be deployed to support research that has the greatest potential to improve patient treatment and care.
What this means:
We will lead and take part in well-designed, high-quality Clinical Trials and other research studies, providing the evidence base required to bring new, improved treatments and interventions into the clinic to enhance patient care. We will prioritise research that is led from Wales. Work in this Theme (and Theme 1) will increase opportunities for patients to participate in research so that within 10 years most of our patients will have the opportunity to take part in research and/or innovation at some point in their cancer journey. Targets for each sub-theme within this theme are specified below.
In this theme we will:
-
- Increase the number of new studies led by local researchers
- Widen access to Late Phase Clinical Trials and deliver these to time and target
- Provide enhanced access to Early Phase Clinical Trials and Solid Tumour Advanced Therapies
- Maximise opportunities for Radiotherapy Research afforded by investment in new VCC
- Integrate Novel Imaging Research into our Clinical Studies
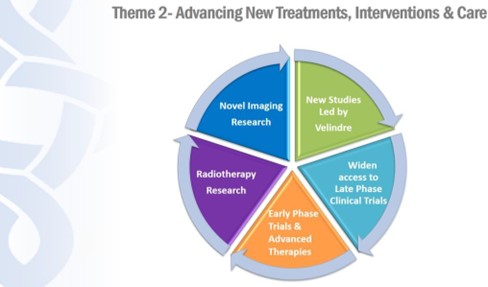
Figure 6: Advancing new treatments, interventions and care
What we will do to achieve this:
Increase the number of new studies led by local researchers by:
-
- Building the future generation of medical and non-medical Chief Investigators (CIs) to lead these studies. We will invest in the best, upskilling, mentoring and developing our future research leaders and ensuring Velindre representation on key national research groups.
- Supporting our researchers with grant and protocol writing/ethics submission and study set-up through new research office function (see Theme 4).
- Systematically looking to overcome the barriers to becoming a clinical research lead in cancer in the NHS in Wales identified in a recent (2020) survey conducted by the Centre for Trials Research (CTR, CU), Research Design and Conduct Services (CU) and WCRC, including: lack of time (identified by 91% of respondents); process and knowledge barriers (e.g. how to build a research team and identify funding opportunities) and systemic barriers preventing cross-organisational research (e.g. between the University and NHS).
Target: to double the number of local Chief Investigators within 10 years, from the current baseline of 18 (see Appendix 3 and 4A) to >30.
Widen access to Late Phase Clinical Trials and deliver these to time and target by:
-
- Adopting a portfolio of trials across all tumour sites so that the vast majority of patients with a cancer diagnosis will have an opportunity to participate in a clinical trial.
- Assessing all new trial opportunities with a robust selection tool that will optimise a broad portfolio of high quality studies likely to recruit to time and target with minimal overlap in patient recruitment characteristics
- Linking more closely with Health Board and Academic colleagues to deliver multidisciplinary cancer trials which optimise all facets of research outcomes, including surgical, palliative care and translational research and streamlining R&D functions.
- Developing a mixed portfolio of commercial and investigator led trials that allows high impact research output alongside a sustainable financial model with commercial income being reinvested into R&D activity.
- Balancing our portfolio of interventional trials with observational and qualitative studies led by the multi-disciplinary workforce.
- Bringing research opportunities to patients across South East Wales and where possible deliver the research close to patients’ homes.
- Funding research time for a small number of motivated clinicians, particularly to those generating significant commercial income, so that they can dedicate appropriate time to being Principal Investigators (PIs) to satisfy Contract Research Organisation/Sponsor requirements and also so that they can develop their own research expertise.
- Embrace research from all clinical disciplines (medical and non-medical).
Target: to ensure that within 10 years most of our patients are involved in research at some point in their cancer journey.
Provide enhanced access to Early Phase Clinical Trials by:
-
- Developing an integrated working plan with our Haemato-oncology colleagues and driving plans to deliver Early Phase Trials across Velindre and UHW sites as per the Nuffield recommendations (see below*1 and Section 5).
- Advancing collaborations with Cardiff University scientists, ECMC and WCRC to deliver at least one bench to bedside project with a Cardiff generated molecule/therapy.
- Designing and delivering 4 investigator-led Early Phase Clinical Trials with Cardiff Chief Investigators through interaction with Industry, Academic colleagues, CRUK and Cancer Research Wales.
- Developing a broad, well-balanced Early Phase Clinical Trial portfolio including both commercial and academic investigator-led studies, aiming to have 15-20 studies open to recruitment at any one time (an approximate doubling of current numbers).
- Expanding our portfolio of Early Phase Drug-Radiotherapy Combination Clinical Trials and joining a network with other UK centres being currently set- up to deliver these.
- Re-investing a proportion of the income generated to provide sustainability of existing posts, an expansion of an expert workforce and infrastructure.
- Contributing to the global understanding, and build on local clinical expertise when these new treatments become available as part of NHS standard care.
Target: to double patient recruitment to Early Phase Trials within 10 years, from a baseline of 25 patients per annum (average annual recruitment 2015-2020, range 17-36 patients) to over 50 patients per annum.
*1The primary endpoint of ‘first-in-human’ Phase I trials is safety and given the changing landscape of Early Phase Trials towards testing immuno-oncology and other intravenous drugs, either alone or in combination, the majority of future Early Phase Trials for solid cancer will require delivery on an acute hospital site with direct access to HDU and ITU. This is recognised in the Nuffield Trust report which recommends the development of a ‘strong research hub’ at UHW to enable Phase I trial delivery and closer working with haemato-oncology, other services and Cardiff University colleagues. Advanced Therapies will also need to be delivered on an acute site and the infrastructural and workforce requirements for their delivery must be considered alongside our future plans for Early Phase trial delivery. Although the majority of these trials will require delivery on an acute site, some ‘lower risk’ studies may be suitable for delivery at the nVCC and a robust process for identifying these studies will be developed.
Provide access to Solid Tumour Advanced (Cell and Gene) Therapies by:
-
- Seeking opportunities to lead with our partners (particularly at C&V UHB) the delivery of Clinical Trials to test the benefit of Advanced Therapies in solid cancers (involving gene therapy, cell and tissue based therapies and tissue engineered products) for the population of South Wales.
- Learning from the experience and expertise of our haemato-oncology colleagues of delivering Advanced Therapies (e.g. CAR-T therapy) in haematological malignancies and supporting closer working and co-localisation to expedite necessary knowledge transfer and delivery capability.
- Investing in the infrastructure and workforce required to deliver these therapies as part of our Early Phase Trials delivery team.
- Joining a collaborative network of R&D delivery teams across the UK to share experience and best practice in this emerging field of research.
- Pro-actively seeking out and developing links with academic and commercial developers to partner with in advanced therapy clinical translation and trial delivery.
- Utilising the knowledge gained in delivering Advanced Therapies in a trials setting to facilitate and expedite their adoption and equitable availability as routine standard of care when fully licenced and commissioned.
Target: Within 2 years – to open at least one solid tumour non-cellular advanced therapy trial (e.g. an oncolytic virus) and one cellular advanced therapy trial annually. Within 5 years – to open two solid tumour non-cellular and cellular advanced therapy trials annually. Within 10 years – to open 5 solid tumour non-cellular and up to 5 cellular advanced therapy trials annually.
Maximise opportunities for Radiotherapy Research afforded by investment in nVCC by:
-
- Ensuring RD&I engagement in the procurement process for the Integrated Radiotherapy Solution (IRS) and driving plans and opportunities for the Radiotherapy Research Bunker associated with nVCC (see below*2).
- Providing opportunities for Welsh patients to access Proton Beam Therapy (PBT) Clinical Trials, by collaboration with the Rutherford Cancer Centre (RCC) and HCRW; working with UK collaborators to develop future PBT trials for a range of cancers (Brain/Oesophagus/ Lung/Head and Neck).
- Expanding the multi-disciplinary radiotherapy research workforce, enabling non-medical (e.g. radiographers) as well as medical researchers to lead future radiotherapy studies.
- Supporting Medical Physics research in advanced technical radiotherapy e.g. to evaluate the potential of automated planning via EdgeVCC (Experience Driven plan Generation Engine by Velindre Cancer Centre) to increase the quality and efficiency of planning and facilitate RTTQA across the UK.
- Collaborating with CU School of Engineering on artificial intelligence (AI), radiomics and image analysis research e.g. to further develop ATLAAS (Automatic Decision Tree Algorithm for Advanced Segmentation) a machine learning tool to delineate cancers on PET-CT scans.
- Establishing an IT infrastructure (‘CardiffCAT’ – Computer Aided Theragnostics) within the NHS in Velindre linking routine NHS clinical data with radiotherapy planning, RTTQA and image analysis data to better predict and understand treatment response.
Target: to design and deliver at least one research study with a radiotherapy focus per annum for the next 10 years.
*2The investment in nVCC will include a fleet of new, cutting-edge Linear Accelerators (LINACS) and a new Integrated Radiotherapy Solution (IRS) with innovative workflow processes which will streamline radiotherapy pathways, increasing efficacy and treatment quality. A Radiotherapy Research Bunker, which (pending funding) will accommodate a dedicated radiotherapy research machine and significantly increase our capacity and capability to conduct cutting-edge radiotherapy research. Developing and expanding Medical Physics R&D will be crucial to fully exploit research and innovation opportunities arising from the IRS in 5 years+ time, as will developing radiographer research capacity enabling integration of patient focused research into every radiotherapy study.
Integrate Novel Imaging into our clinical studies by:
-
- Making greater use of local imaging expertise and the state-of-the-art imaging facilities at CUBRIC and PETIC to develop internationally-competitive research in cancer imaging.
- Developing a programme of local investigator-led innovative imaging research studies, incorporating both smaller standalone hypothesis-generating research studies and studies embedded within translational components of larger research trials that are led from Wales. These studies, led by medical or non- medical researchers, will aim to:
- Improve precision of tumour delineation prior to starting a course of radiotherapy
- Image tumours during a course of radiotherapy and adapt radiotherapy treatment according to early markers of tumour response
- Accurately verify tumour and patient position during radiotherapy
- Image tumours after a course of radiotherapy to more accurately define tumour response to treatment
- Study the effect of radiotherapy on normal tissues.
- Integrate novel imaging into the everyday radiotherapy patient pathway, thereby increasing participation in research and learning from every patient.
- Collaborating with partners at the CU School of Engineering to further develop the SPARCC (Automate Analysis and Radiomics Computing) pipeline, using data from medical images to tell us about disease behaviour and/or treatment response (radiomics).
Target: to design and deliver at least one research study with an imaging focus per annum for the next 10 years.
Further details our radiotherapy and imaging research plans are included in the VUNHST Radiotherapy (RT) Research Strategy 2020-2025. We will establish leadership for each research domain in this theme, develop the multi-disciplinary research workforce (to include medics, physicists, radiographers, research fellows) and work with CU collaborators (e.g. for imaging, through establishing on-site presence at CUBRIC and/or PETIC for 1 day/week), mentor the next generation of researchers and develop an infrastructure to support our research (see Theme 4).
4C. Theme 3: Driving translational research through connecting the laboratory and the clinic
We will develop a portfolio of clinical and technical translational research, focusing where appropriate, on a few key tumour sites so that we are nationally competitive and can attract inward investment in these areas.
What this means:
Velindre has a key role to play in translating new research findings from the laboratory (the ‘bench’) to the clinic (the ‘bedside’) for patient benefit and encouraging a culture change around research translation for the benefit of health as highlighted in CRUK’s report ‘Bench to Bedside: Building a Collaborative Medical Research Environment in Wales’ (16). This includes translation of new technological discoveries into the clinic, as well as laboratory research. Translational research cannot happen without close and collaborative working between academic researchers (laboratory, pre-clinical or basic scientists) and clinical researchers in the NHS and on developing posts which link across the clinical-academic boundaries. Working with CU and other academic partners we will be part of a shared and coordinated focus, fostering areas of excellence in which we can compete for funding with the best researchers in the UK and internationally. In recent years, Cardiff’s bids to renew CRUK Centre status (2017) and become a CRUK Radiation Network (RadNet) Centre of Excellence (2019) were unsuccessful. Feedback from CRUK on both bids highlighted a lack of infrastructure and critical mass of cancer researchers in Cardiff which must be addressed in order to build research excellence and make us (and Wales) nationally and internationally competitive to attract future investment.
Translational ‘bench to bedside’ research can include bringing novel agents, developed in the laboratory, to the clinic to assess their safety and activity in patients (Figure 7). It can also involve bringing new imaging techniques and/or technological developments from academia into the clinic. ‘Reverse translation’ (‘bedside to bench’) research is where patient samples are taken to the laboratory to generate new knowledge (that can subsequently be ‘translated’ back to the clinic e.g. in the form of new treatments) and it is also crucially important and Velindre has a critical role to play here.
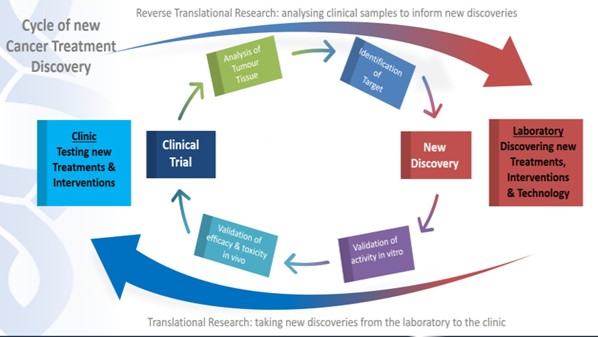
Figure 7: Driving translational research through connecting the laboratory with the clinic
In this theme, we will:
-
- Develop a nationally competitive portfolio of translational research with our partners
- Develop career pathways for clinical academic and other academic-NHS ‘interface’ posts
- Improve dialogue and collaboration between the NHS and academia
- Be a partner in regaining CRUK Centre Status in Cardiff
-
- What we will do to achieve this:
Develop a nationally competitive portfolio of translational research with our partners by:
-
- Working closely with academic researchers within the Cancer Theme at CU to identify areas of strength in basic cancer research.
- Linking-up clinical researchers within the NHS at Velindre and basic science researchers at CU to ‘bridge the translational gap’ between the laboratory and clinic.
- Mapping out a clear path to translation of scientific discoveries in the laboratory, linking basic science to the clinic by focusing on translation and developing a pipeline to bring novel therapies from the laboratory through to the clinic for testing in Early Phase Trials.
- Utilising fully the unique infrastructure for translational imaging research available in Cardiff and Wales, including the CUBRIC and chemistry expertise at Wales’ PET Imaging centre.
- Increasing the translational richness of all our Clinical Trials and maximising reverse translational opportunities in every study as they are conceived.
Develop career pathways for clinical academic and other academic-NHS ‘interface’ posts by:
-
- Supporting research leaders by creating defined clinical academic, nurse/AHP researcher, healthcare scientist career pathways.
- Supporting early career researchers in medical and non-medical disciplines at MSc and PhD level, mentoring them towards Fellowships (targeting prestigious MRC, CRUK and NIHR awards) and supporting their development to become future research leaders (see Theme 4).
Increasing the number of honorary academic appointments. We support the following statement in the Academy of Medical Science report in 2020 (17): “academic HEIs should increase number of honorary academic appointments for NHS staff who contribute significantly to research (evidenced in job plans), recognise contribution in REF & allow access to: grant-making machinery, career development, mentoring, training, promotion opportunities; and opportunities for student supervision”.
Improve dialogue and collaboration between the NHS and academia by:
-
- Encouraging staff to join the WCRC’s Multidisciplinary Research Groups (MDRGs) and other fora through which the ideas of academic researchers can be heard and facilitated and to bring academic researchers closer to cancer patients’ priorities and opportunities for research.
- Improving our links and representation with national research organisations such as Medical Research Council (MRC), National Institute for Health Research (NIHR), National Cancer Research Institute (NCRI) and UK Research and Innovation (UKRI).
Help to regain CRUK Centre Status in Cardiff by:
-
- Strengthening multidisciplinary academic collaborations, bringing our clinical expertise, researchers and research proposals to consolidate a strengthened cancer research critical mass that enables the expansion of research excellence and attracts inward investment.
- Working to support other important bids, including Cardiff’s ECMC application.
4D. Theme 4: Embedding research and innovation within the organisational culture and structure
We will establish an organisational culture which prioritises, values and supports research for the long term. It is important to note whilst our major focus will be clinical research, we recognise the value of other research (such as Health Services, Implementation and Operational research) that will be conducted to improve our services. To embed all research in and across organisational structures we will develop a unified governance process for clinical and non-clinical research to oversee these activities enabling better cross linking of research and prioritising Trust resource allocation.
What this means:
The Welsh Government is committed to creating a healthcare system that truly values research across NHS Wales. Our Board like that of every NHS Trust, has responsibility for valuing and promoting research across the organisation. Research is good for patient care but there are barriers to greater adoption of research into day-to-day service which may, in part, be because research success has traditionally been portrayed in very academic terms and it is not the currency the NHS recognises. In contrast, patient care and patient benefit is the currency of success for a healthcare worker and healthcare organisation. As a consequence, NHS staff may not consider research to be part of their remit, however, because of the strong relationship with patient care, it is totally the business of everyone working in the NHS at Velindre. This theme ensures research feeds into the mechanisms for the uptake of best practice, innovation and service change and that service changes and the impact on patient care are evaluated and shared.
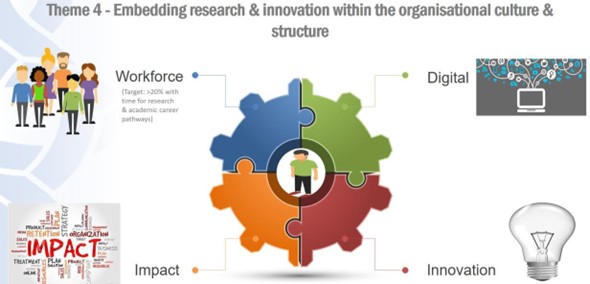
Figure 8: Embedding research and innovation within the organisational culture and structure.
In this theme we will:
-
- Establish a culture that values research and every member of the research workforce.
- Build capacity, capability and a critical-mass within the research workforce
- Create a supporting infrastructure to underpin our research.
- Innovate to implement new knowledge into clinical practice.
- Demonstrate the impact of our research on patient care and the NHS in a systematic way.
What we will do to achieve this:
Establish an organisational culture that truly values research by:
-
- Increasing the understanding of research and its benefits across the organisation, dispelling the perception that research is only carried out by certain staff.
- Celebrating research success across the organisation and promoting our work through communication and engagement with patients, staff and partners.
- Engaging with plans for the ‘Centre for Learning’ in the nVCC, fostering partnerships that can benefit research and innovation.
- Improving our integration in academic health science partnerships such as all Wales Multidisciplinary Research Groups (MDRGs) and inter-professional research sandpits that stimulate internal cross fertilisation and synergy in research approach.
Build capacity, capability and a critical-mass within the research workforce by:
-
- Providing opportunities for NHS staff of all disciplines to develop research skills, ensuring research informed practice is a core principle across the multi- professional workforce and incorporating research professional development into staff education and workforce development strategies.
- Providing dedicated, ring-fenced research time for NHS staff (Medics, Physicists, Radiographers, AHPs, Healthcare scientists, Nurses, Pharmacists, Psychologists and other staff) which is formally acknowledged in job plans to build capacity and a robust and sustainable multi-disciplinary workforce. We will aim to fund 20% of the Velindre workforce to dedicate 20% of their time to research within 10 years.
- Enabling more clinical researchers to be Principal Investigators (PIs), building confidence and understanding and allowing time in job plans for this important activity which is critical to expand our portfolio of clinical and supportive care research.
- Ensuring succession planning across the clinical workforce to ensure research activity is future proofed.
- Allowing clinical research staff to work flexibly across organisational boundaries, ‘following the patient’ to provide research opportunities for patients across SEW.
- Supporting early career researchers in medical and non-medical disciplines at MSc and PhD level, mentoring them towards Fellowships (targeting prestigious MRC, CRUK and NIHR awards) and supporting their development by ensuring a Clinical Research Fellow career pathway for them to become future research leaders.
- Working closely with our academic partners, we will target talented researchers and enable the Clinical Academic career pathway to build critical mass in certain areas and establish our future research leaders (in medical and non- medical disciplines). We will invest in the best, developing business cases to support research time for targeted individuals, with a view to establishing tenured academic posts for those who fulfil pre-determined university metrics (for research and teaching) within 5 years. We will aim to identify and support at least 1 talented individual per annum along the Clinical Academic career pathway, in collaboration with our University partners.
Create a supporting infrastructure to underpin our research by:
Creating a supporting digital infrastructure:
-
- Embracing opportunities for research and innovation afforded by the NHS digital transformation. Digital healthcare technologies are defined in the Topol review 2019 as genomics, digital medicine, artificial intelligence (AI) and robotics and a number of emerging technologies, including low-cost sequencing, telemedicine, smartphone apps, biosensors for remote diagnosis and monitoring, speech recognition and automated image interpretation, will be particularly important for the healthcare.
-
- Creating a digital infrastructure and a capacity within the digital workforce to enable clinical research activities. This includes linking clinical researchers with nominated members of the Trust’s digital team and developing a ‘Living platform for research’ which we envisage will be a virtual ‘repository of ideas’ accessible to researchers across the Trust as a forum to discuss of new research ideas and to link researchers together.
- Enabling clinical researcher to exploit the potential of ‘Big Data’ obtained from radiotherapy, chemotherapy, radiology, clinical sources to research cancer outcomes at scale in order to make every patients’ experience count. Furthermore, to interrogate secure data held in the National Data Resource (NDR) to study All Wales outcomes and trends, as per the Welsh Government ‘A Healthier Wales: our Plan for Health and Social Care’.
- Engaging with Health Technology Wales (HTW), hosted by VUNHST, to appraise new developments and innovations.
Creating a research infrastructure that enables research delivery and development of new research:
-
- Establishing a strategic leadership team representing the research themes to drive the research and innovation agenda, ensuring oversight of alignment, partnerships, investment and performance.
- Transforming our research portfolio processes, optimising resource utilisation, prioritisation, financial transparency and efficiencies to widen research participation.
- Ensuring our research delivery workforce has capacity and is appropriately skilled, experienced, and agile to deliver high quality research support and patient care.
- Support the development of new research by providing practical support and advice for staff (such as research costings, study design, research approval) and transparent signposting pertinent information and funding opportunities to new researchers.
- Research is becoming increasingly complex to set-up and deliver, because of the associated trial regulation, legislation and paperwork which is made more cumbersome when working across organisational boundaries. Streamlining processes across the network by alignment of R&D offices and procedures should be explored, learning from the Joint Research Office initiative already established between C&V UHW and CU.
Innovate to implement new knowledge into clinical practice by:
-
- Using innovation as a key enabler for NHS Wales to deliver ‘A Healthier Wales’…” Linking research seamlessly with innovation to ensure that new knowledge is implemented into clinical practice as set out in the NHS Wales Planning Framework 2020-23. Innovation “should be organisationally aligned with and follow research, translating new knowledge into better practice.”
-
- Working with others, particularly universities and industry partners, must be a key part of our approach to innovation.
- Developing an Innovation strategy for VUNHST to align with this R&D ambition document and other organisational and regional priorities for service improvement etc.
Demonstrate the impact of our research on patient care and the NHS by:
-
- Collecting information on patient outcomes, patient care, service delivery and cost-effectiveness in a comprehensive and systematic way, including data on all grant awards and all publications by our researchers.
- Increasing research income – through grant income, commercial partnerships and fundraising.
- Publicising our research and research findings on the Trusts’ webpages, social media and other platforms.
As well as the culture and infrastructure in this theme, partnership working will be key to our success (see Section 5). We will develop effective and mutually beneficial R&D partnerships with our regional UHBs and Academic institutions and seek out diverse strategic collaborations across different sectors (Patients, Public, NHS Wales, Academia and the Third Sector) for research benefit. We will look to work with a range of industry and commercial sectors (commercial trials, diagnostics, medical technology), as well as with associated industries such as digital, design and artificial intelligence. We will develop ongoing dialogue with all partners reporting on our research work where research investment has been made, seeking partner feedback to improve our partnership approaches.
5. The Nuffield Advice and Future Model for Research and Development
5A. The Nuffield Advice
Our current service model and a future stand-alone new Velindre Cancer Centre, in isolation, do not enable the full range of research ambition described in this document. In particular:
- Based on current data, we estimate that >50% of future Early Phase Clinical Trials, which test the safety of novel systemic therapies and most trials of solid tumour Advanced Cellular Therapies and vaccines can only be safely delivered on an acute hospital site, where the ability to rapidly escalate care to on-site HDU/ITU and to access other specialist services to manage treatment toxicities (e.g. gastroenterology, cardiology, immunology) is available. Furthermore, opportunities to share research facilities and staff with other cancer disciplines (Haemato- oncology and Teenage and Young Adult [TYA] services) cannot be realised.
- It is challenging to translate basic research, undertaken by academic scientists in the laboratory at CU, into the clinic for patient benefit. Collection of bio-resources to underpin research programmes, includes taking biopsies of patients’ tumours (as well as blood/saliva etc.) which requires surgical and interventional radiology services, supported by on-site HDU/ITU.
- Expanding our portfolio and providing more opportunities for patients to take part in research requires facilities closer to their homes.
The landscape of emerging systemic treatments has changed dramatically over the last five years from relatively safe oral small molecules to higher risk intravenous immune checkpoint inhibitor therapies. This trend is likely to continue for the foreseeable future with the emergence of Advanced Cell and Gene Therapies, meaning that patients will be increasingly unable to participate in cutting edge research on a stand-alone site. Cancer research in general has changed too, with large-scale infrastructural funding (e.g. from CRUK for CRUK Centres, ECMCs, RadNet centres and CTRs) becoming concentrated in fewer institutions with a ‘critical mass’ of cancer researchers and well-developed research infrastructure capable of undertaking a wide range of academic research and clinical trials of new cancer treatments. Establishing this infrastructure in Cardiff is essential to attract investment, build capacity and expertise, and ensure that research becomes seamlessly integrated into clinical care to improve outcomes for the population of SEW.
The Nuffield Trust report (Dec 2020) states that “successful research is a key element of high-quality cancer provision”. It acknowledges the limitations to our current service model and provides solutions to overcome these limitations in the future, in the context that a new Velindre Cancer Centre is built close to its existing site and that full co- location of the new Velindre Cancer Centre on CU/UHW site is not feasible, at least for the next 15 years. It concludes that there is a need for a “strong research hub at UHW and also at other hubs across the network”.
Section 5 of the Nuffield Trust report ‘Building research excellence’ makes the following recommendations:
- “An agreed research strategy is clearly a priority. This needs to include research in its widest definition, including research led by disciplines other than medicine.
- There is more work to do to make the network model work well and in particular to remove some of the governance and bureaucratic barriers to research across sites and LHBs.
- There is a close alignment between the strategy for cancer services, the development of the research network and our recommendation for a research hub at UHW* to be developed alongside the enhanced Velindre-supported AOS. This should work closely with the haemato-oncology service and include much better-coordinated working with other specialties. This would enable Phase 1 trials to take place at UHW that require ITU support and also other Phase 1, 2 and 3 trials at the VCC and in Velindre@ locations. Phase 1 trials are important but the capability to do a wide range of trials across the network is even more so.
- The other Velindre@ units in LHBs need to be viewed as a key part of the research delivery network** and supported accordingly as they also have access to large numbers of patients and support from ITU and other specialties.
- One of the experts we spoke to would encourage this group to ensure it has benchmarked the research approach and capabilities with other comparable research networks.
- Some members of the research group have a preference for the location of the radiotherapy research bunker to be at UHW rather than at the new VCC. However, opinions differ and there is an efficiency penalty for this. There are some emerging research areas in which immunotherapy is combined with radiotherapy, which might suggest that location should be considered. However, given the uncertainties around the future of the UHW site, practical obstacles and the efficiency penalty, we suggest that at present the linear accelerators should be provided in a single bank at VCC with arrangements made to transport patients or research staff where required”.
*In addition to synergies with other clinical disciplines the report states that the research hub at UHW “offers opportunities for closer working with the university, which are going to be increasingly important in a number of areas as the need for multidisciplinary research expands.”
**The report suggests that a strong research network across the whole of SEW will (based on existing literature) require “the following common elements:
- a shared vision
- formal governance policies and terms of reference
- an infrastructure team dedicated to the goals and activities of the network
- regular and effective communication
- a framework for holding members to account
- a succession planning strategy to address membership change over time
- multiple strategies to engage network members
- regular reviews of goals and timelines
- a balance between structure and creativity.
Clearly, there is a lot of work to do to ensure that some of these elements are in place”.
5B. A future model for cancer R&D across South East Wales:
Based on the Nuffield recommendations, we envisage the following regional model for cancer R&D across South East Wales:
- A new Velindre Cancer Centre which is designed to enable R&D with the workforce capacity and capability to make research widely available to patients attending the centre. It is envisaged that R&D supporting infrastructure will be located within the proposed Centre for Learning (C4L) at nVCC, co-located with innovation. Clinical research including ‘low risk’ phase II and III Clinical Trials, Supportive and Palliative Care research led by the multi-professional non-medical and medical research workforce, including Medical Physics and radiotherapy research will be undertaken here. Developing the portfolio of radiotherapy research to optimise opportunities afforded by the new fleet of radiotherapy LINACS and making the case for the radiotherapy research bunker will be a priority.
- A Cardiff Cancer Research Hub on CU/UHW C&VUHB site which will bring together cancer patients, NHS cancer researchers (from C&VUHB and Velindre) and academic cancer researchers (from CU School of Medicine) in one location. This tripartite hub will provide a focus to join-up cancer research in Cardiff, invigorate the cancer research community and provide facilities for:
- Delivery of Early Phase Trials and Advanced Cellular Therapies for solid cancer with access to HDU/ITU and specialist services (eg surgery, cardiology, immunology, gastroenterology) to manage the complications of therapy and enabling “collaboration with haemato-oncology research” and Teenage and Young Adult (TYA) services.
- Translational research by enabling “closer working with the university”, including clinic, office and meeting space for basic (academic) and clinical researchers, with direct links to the laboratory, biobank, surgery and interventional radiology.
- An enhanced, integrated, multi-disciplinary Clinical Academic workforce, responsible for designing and delivering cancer research that is led from Wales.
The hub will work closely with other cancer disciplines (TYA oncology, haemato- oncology, medical genetics) to share facilities and staff where that is the best model. It would also be ideally located alongside an Acute Oncology facility, with mutual benefits, so that Clinical and Research Models could be integrated. It will provide potential advantages to training and education from being co-located with the School of Medicine, attracting the attention of medical, nursing and other students and inspire multi-disciplinary staff groups to take an interest in oncology early on in their careers.
- Velindre@ Research Facilities at Aneurin Bevan UHB (including the Radiotherapy Satellite at Nevill Hall), Cwm Taf Morgannwg UHB, as well as within the Cardiff Cancer Research Hub at CVUHB, forming a multicentre clinical research network across SEW, which will be critical to increase opportunities for patients to access research across the region, through a ‘hub and spoke’ mode. It is envisaged that these will be centres for delivery of Phase II-III Clinical Trials, Supportive and Palliative Care studies led by the multi-professional non-medical and medical research workforce and (at the Radiotherapy Satellite) Radiotherapy studies.
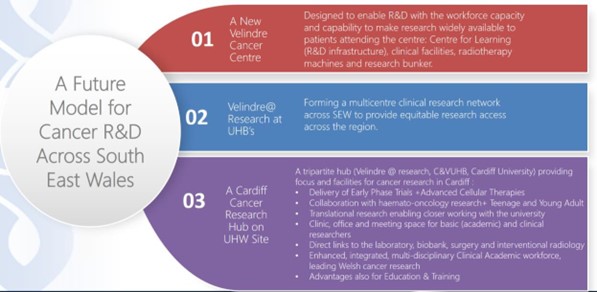
Figure 9: A future model for Cancer R&D across South East Wales
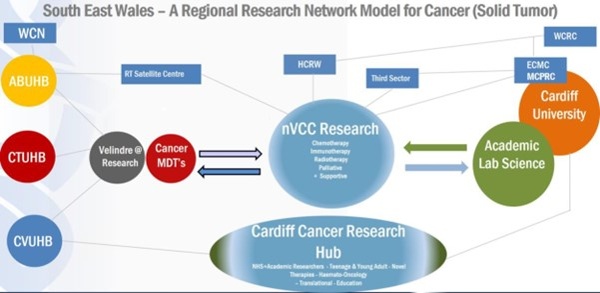
Figure 10: South East Wales - A Regional Research Network for Solid Tumour Cancer Research
5C. Delivering the Service Model:
Establishing the R&D model described above will require people, funding and infrastructure, as well as close working with UHBs and Academic partners across the SEW region. There will be elements that will need to be delivered internally by VUNHST via the ongoing Velindre Futures programme. There will be others that will need to be delivered in partnership with CU via a refreshed Velindre-Cardiff University Partnership Board with the remit to develop a joint strategic research partnership. Finally, there will be elements that will need to be delivered regionally with our UHB partners (C&VUHB, ABUHB and CTUHB) and, as well as infrastructure, consideration will need to be given in the detailed implementation plan of how the multi-disciplinary research workforce will work across organisational boundaries to deliver research studies close to patients’ homes. This will be facilitated through the regional SEW Cancer Collaborative Leadership Group (CCLG), which is complementary to the national Wales Cancer Implementation Group (CIG) with delivery and implementation responsibilities for the SEW region. Currently, plans for a Regional Cancer RD&I Group to be set-up under the overarching CCLG structure are being developed. It is envisaged that this could be the key group responsible for improving collaboration between NHS Wales organisations to enable successful cross-organisation working and turn the regional ambitions and proposed delivery model in this paper into detailed plans which can be incorporated into the future TCS Programme Business Case and delivered across the SEW region.
6. Next Steps and Conclusions
6A. Next Steps
- Consultation and Engagement
We will engage with Velindre staff, our University Health Board and Academic partners to share the ambitions laid out in this document. Thereafter, we will engage with Charities, Industry and the Welsh Government who will be key partners to ensuring cancer research delivery, sustainability and growth across the region and beyond.
- Benchmarking
Benchmarking (as recommended by Nuffield) with 4 UK Cancer Centres across both strategic and operational management approaches to R&D to identify key areas, infrastructure, service models, factors and processes and that contribute to the Centres’ R&D success.
Aims of benchmarking will be to:
-
- Inform VCC’s strategic thinking and operational planning for the next decade
- Share best practices, enabling opportunities for adoption of new processes/ new ways of working, reducing duplication of effort.
- Provide better understanding to explain differences in performance data
- Promote VCC’s R&D capability, strengths and interests with the aim of future research collaboration and creating ongoing informal alliances allowing periodic benchmarking.
- Detailed Implementation Plan with Timelines
A detailed implementation plan will need to be set-out, in discussion with our UHB and Academic partners, HCRW and others, detailing how these ambitions and Nuffield recommendations for R&D will be realised. It will need to include key milestones and key performance indicators across all areas of research portfolio including:
-
- Infrastructural requirements for a Cardiff Cancer Research Hub and Velindre@ Research Facilities at each UHB across SEW.
- Description of the research network required to deliver research to patients across SEW.
- Requirements for developing a multidisciplinary research workforce within the NHS as well as developing clinical academic career pathways for research leaders with Cardiff University and other academic partners.
- Short (what can be done now), medium (in 5 years’ time) and longer-term plans (in 10+ years) for implementation for each of the key research themes in this document.
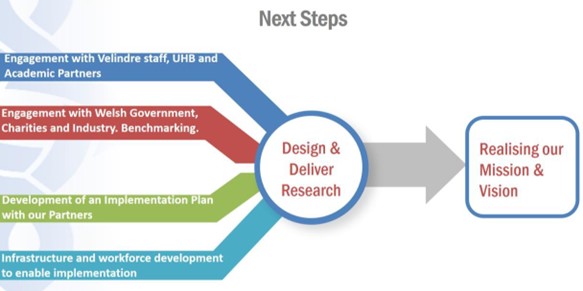
Figure 11: Next Steps
6B. Conclusions
The ambitions and strategic priorities laid out in this paper, if delivered, will not only develop long-lasting partnerships which will drive and enable cancer research, they will bring forward benefits for cancer patients (now and for the future) across SEW. The network model will require new infrastructure, regional collaboration between the Trust and UHB partners and a joint strategic research partnership between the Trust and Cardiff University which will, if realised, accelerate new discoveries into the clinic and build a critical mass of cancer research activity in SEW that will generate inward investment and growth, enabling research success and with it research sustainability.
7. References
- Downing A, et al. (2016). High hospital research participation and improved colorectal cancer survival outcomes: a population based study. Gut 66, 89-96.
- Nijjar SK, et al (2017). Participation in clinical trials improves outcomes in women’s health: a systematic review and meta-analysis. BJOG. DOI: 10.1111/1471-0528.14528
- https://www.nihr.ac.uk/documents/research-participant-experience-survey-report-2018-19/12109 (Accessed 20 Jan 2021)
- Ipsos MORI (2016). Public Support for research in the NHS.https://www.ipsos.com/ipsos-mori/en-uk/public-support-research-nhs
- Jonker L & Fisher SJ (2018). The correlation between National Health Service trusts’ clinical trial activity and both mortality rates and care quality commission ratings: a retrospective cross-sectional study. Public Health 187, 1-6.
- Boaz A, et al. (2015). Does the engagement of clinicians and organisations in research improve healthcare performance: a three-stage review. BMJ Open 5, e009415.
- Rees MR, Bracewell M, Medical Academic Staff Committee of the British Medical Association (2019). Academic factors in medical recruitment: evidence to support improvements in medical recruitment and retention by improving the academic content in medical posts. Postgraduate Medical Journal;95(1124):323-327. DOI: 10.1136/postgradmedj-2019-136501
- https://gov.wales/sites/default/files/publications/2019-09/nhs-wales-planning- framework-2020-to-2023.pdf
- Crosby, T., C. N. Hurt et al (2013). Chemoradiotherapy with or without cetuximab in patients with oesophageal cancer (SCOPE1): a multicentre, phase 2/3 randomised trial. Lancet Oncology 14(7): 627-637.
- Warde P, Mason M, (JOINT FIRST AUTHORS), et al (2011). Combined androgen deprivation therapy and radiation therapy for locally advanced prostate cancer: a randomised, phase 3 trial. Lancet 378 (9809):2104-2111 DOI: 10.1016/S0140-6736(11)61095-7
- Owadally W….Evans M (2015). PATHOS: a phase II/III trial of risk-stratified, reduced intensity adjuvant treatment in patients undergoing transoral surgery for HPV-positive oropharyngeal cancer. BMC Cancer 602.
- Jones R, Casbard A et al (2020). Fulvestrant plus capivasertib versus placebo after relapse or progression on an aromatase inhibitor in metastatic, oestrogen receptor- positive breast cancer (FAKTION): a multicentre, randomised, controlled, phase 2 trial. Lancet Oncology 21(3):345-357.
- Wheeler PA, Chu M, Holmes R, Smyth M, Maggs R, Spezi E, et al. Utilisation of Pareto navigation techniques to calibrate a fully automated radiotherapy treatment planning solution. Phys Imaging Radiat Oncol 2019;10:41–8. doi:10.1016/j.phro.2019.04.005.
- Roberts R, Borley A et al (2020). Identifying Risk Factors for Anthracycline Chemotherapy-induced Phlebitis in Women with Breast Cancer: An Observational Study. Clinical Oncology. https://doi.org/10.1016/j.clon.2020.11.025
- Ball S, et al. (2019). Patient and public involvement in research: enabling meaningful contributions. https://www.rand.org/pubs/research_reports/RR2678.html
- https://www.cancerresearchuk.org/sites/default/files/bench_to_bedside_wales_full_re port.pdf
- https://acmedsci.ac.uk/file-download/23932583
8. Appendices
Appendix 1. Authorship Group
|
Name |
Title |
Institution |
|
Prof Mererid Evans CHAIR |
Associate Medical Director of R&D |
VUNHST |
|
Dr Jacinta Abraham |
Executive Medical Director / Lead for Research Development and Innovation |
VUNHST |
|
Mrs Libby Batt |
Research, Innovation and Improvement Co- ordination Lead |
VUNHST |
|
Dr Robert Jones |
Clinical Director of R and D VCC |
CU/VCC |
|
Dr Mark Briggs |
Head of Cell & Gene Therapy / Lead for RD&I Strategy |
VUNHST |
|
Prof Jane Hopkinson |
Velindre Professor of Nursing and Interdisciplinary Cancer Care |
CU/VCC |
|
Dr Paul Shaw |
Director for Radiotherapy Research |
VCC |
|
Dr James Powell |
Leading Proton Beam Research |
VCC |
|
Dr Cath Matthams |
Radiotherapy R&D Lead |
VCC |
|
Prof Anthony Byrne |
Consultant in Palliative Care & Medical Director Marie Curie Palliative Care Research Centre |
CU/VCC |
|
Mrs Sarah Townsend |
Head of Research & Development |
VUNHST |
|
Dr Phil Wheeler |
Clinical Scientist- Medical Physics |
VCC |
|
Dr Emiliano Spezi |
Leader, Medical Engineering group and Cancer Imaging and Data Analytics team |
CU |
|
Prof Awen Gallimore |
Cancer College Theme Lead |
CU |
|
Mrs Sophie Harding |
Pharmacy Research Lead |
VCC |
|
Ms Francesca Carpanini |
Engagement and Communications Manager |
VUNHST |
|
Prof Alan Parker |
Professor of Translational Viro-therapies |
CU |
|
Mr Bob McAlister |
Public Representative |
- |
|
Mr Alan Buckle |
Patient Representative |
- |
|
Mrs Sandra Cusack |
Medical Director Personal Assistant |
VUNHST |
|
Mrs Emma Duggan |
Research & Development Secretary |
VUNHST |
[The group would like to thank Ms Laura Tolley (Business Support Officer, Velindre Corporate Services) for her assistance in producing the Figures in this document].
Appendix 2. Aligning our R&D ambitions to National Policies and Strategies.
The list below details strategies, reports and current Velindre University NHS Trust strategies that have informed the Overarching Cancer Research and Development Ambitions and Strategy for Velindre University NHS Trust 2021-31 document.
|
Welsh Policies |
Welsh Strategies |
UK Strategies |
Reports |
Internal |
|
A Healthier Wales -WG 2018.
Well Being of Future Generations Act- WG 2015.
Prosperity for All: the national strategy-WG 2017.
The Reid Review (Review of government funded research and innovation in Wales) on behalf of WG 2018.
End of Life Care Delivery Plan – WG.
Cancer Delivery Plan 2016-2020 –WG. |
CREST – A Cancer Research Strategy for Wales 2020 (pending) WCN/WCRC/W CA Cell and Gene Therapy Statement of Intent WG 2019.
HCRW Support and Delivery Service. 2017- 2022. A Strategic Framework. |
Life Sciences: industrial strategy- A report from life sciences sector (Bell) UK Gov 2017. UK International Research and Innovation Strategy. HM. GOV. 2019.
NCRI Accelerating Cancer Research –A strategy for collaboration between cancer research funders UK (2017-2022.)
The UK Standards for Public Involvement in |
CRUK Bench to Bedside: Building a Collaborative Medical Research Environment in Wales 2019. Transforming health through innovation: Integrating the NHS and Academia. Academy of Medical Sciences. Jan 2020.
The Topol Review - Preparing the Health care workforce to deliver the Digital Future. NHS Constitution 2019. |
VUNHST Invigorate - An Innovation, Education and Life Sciences R and D Strategy.
VUNHST Radiotherapy Research Strategy 2020- 2025. Early Phase Clinical Trials Strategy 2017- 2022.
Transforming Cancer Services (TCS): A Collaborative Centres for Learning, Technology and Innovation Briefing Paper. 2020.
VUNHST Delivering Excellence – |
|
NHS Planning Framework Wales 2020 - 2023 –WG 2019. |
|
Research NIHR 2019. |
|
Integrated Medium Term Plan 2019-2022. |
Appendix 3. Velindre Sponsored Cancer Clinical Studies led by Velindre Chief Investigators (data updated 21-01-2021)
|
Acronym |
Trial Title |
Chief Investigator/s |
|
PATHOS |
A Phase III trial of risk-stratified, reduced intensity adjuvant treatment in patients undergoing transoral surgery for Human papillomavirus (HPV)-positive oropharyngeal cancer https://www.cardiff.ac.uk/centre-for-trials- research/research/studies-and-trials/view/pathos
Funder: Cancer Research UK (CRUK) |
CIs: Professor Mererid Evans and Professor Terry Jones |
|
PEARL |
A prospective, interventional, non-randomised, phase II feasibility study for patients with good prognosis Human Papillomavirus (HPV)-associated oropharyngeal squamous cell cancer (OPSCC) who are suitable for treatment with concurrent chemo-radiotherapy (CCRT). https://www.clinicaltrials.gov/ct2/show/NCT03935672
Funder: Cancer Research Wales (CRW) and Velindre Advancing Radiotherapy Fund |
CIs: Professor Mererid Evans and Dr Thomas Rackley |
|
SCOPE2 |
A Randomised Phase II/III Trial to Study Radiotherapy Dose Escalation in Patients With Oesophageal Cancer Treated With Definitive Chemo-radiation With an Embedded Phase II Trial for Patients With a Poor Early Response Using Positron Emission Tomography (PET)https://www.cardiff.ac.uk/centre- for-trials-research/research/studies-and-trials/view/scope2
Funder: CRUK |
CI: Professor Tom Crosby |
|
STORM_ Glio |
Study of Radiomics in High Grade Glioma
Funder: Engineering and Physical Sciences Research Council (EPSRC) and Velindre Headfirst Brain Tumour Appeal Charitable Fund |
CIs: Dr James Powell and Dr Emiliano Spezi |
|
MRI Mapping of Metabolic |
An integrated MRI tool to map brain microvascular and metabolic function: improving imaging diagnostics for human brain disease. |
CI: Professor Richard Wise |
|
Brain function |
Funder: Engineering and Physical Sciences Research Council (EPSRC). |
Co-investigator: Dr James Powell |
|
iNATT |
interNational Anaplastic Thyroid Cancer Tissue Bank and Database (NATT) https://clinicaltrials.gov/ct2/show/NCT01774279
Funder: Thyroid Cancer Support Group – Wales |
CI: Dr Laura Moss |
|
VIP-Epi |
A study to record the incidence and severity of post epirubicin chemical phlebitis (PECP) and patient reported pain following Epirubicin chemotherapy administered using the Plum 360 volumetric infusion pump. |
CI: Dr Rosie Roberts |
|
Application of VR in an Oncology Healthcare context |
Virtual Reality in an Oncology healthcare context; improving patient experience and increasing treatment acceptance |
CI: Dr Caroline Coffey |
|
ASPIRE |
A study to train and test an artificial intelligence (AI) algorithm to automatically outline oesophageal tumours using computed tomography (CT) and positron emission tomography (PET) scans, using Intel’s advanced computing hardware. Funder: Welsh Government and Intel |
CIs: Professor Tom Crosby, Dr Emiliano Spezi, Dr Kieran Foley |
|
CHROME
(in set-up) |
A study to improve the accuracy of cancer staging by using information from radiology, pathology and genetics to develop a tool that can predict the likelihood of metastatic spread in different tumour types. |
CI: Dr Kieran Foley |
|
SABR_IT |
A study to investigate immune responses following Stereotactic Ablative Radiotherapy (SABR). Funder: Velindre Advancing Radiotherapy Fund (ARF) and Wales Cancer Research Centre (WCRC) |
CIs: Dr Thomas Rackley, Dr Catherine Pembroke, Professor Awen Gallimore |
|
BEST OF |
A randomized phase III study comparing swallowing function after surgery versus radiotherapy in patients with early stage squamous cell carcinoma of the oropharynx. International study led by EORTC, VUNHST is UK Legal Representative
Funder (in UK): CRUK |
CIs (UK): Professor Mererid Evans/Professor Terry Jones |
|
ADVANCE- POCT |
The purpose of this study is to find out if we can use this point of care equipment to improve the management pathway for cancer patients receiving chemotherapy that present with suspected or border-line neutropenic sepsis. |
CI: Professor Richard Adams |
|
CORINTH |
A phase Ib/II trial of checkpoint inhibitor (pembrolizumab an anti PD-1 antibody) plus standard IMRT in HPV induced stage III/IV carcinoma of the anus.https://www.cardiff.ac.uk/centre-for-trials- research/research/studies-and-trials/view/corinth
Funder: MSD |
CI: Dr Marcia Hall and Prof Richard Adams (co-CI) |
|
FAKTION
(closed) |
A Phase 1b/2 Randomised Placebo Controlled Trial of Fulvestrant +/- AZD5363 in Postmenopausal Women With Advanced Breast Cancer Previously Treated With a Third Generation Aromatase Inhibitor https://clinicalrials.gov/ct2/show/NCT01992952https://clinicalt rials.gov/ct2/bye/rQoPWwoRrXS9-i- wudNgpQDxudhWudNzlXNiZip9Ei7ym67VZRFnERF8ER0tA 6h9Ei4L3BUgWwNG0it.
Funder: CRUK and Astra Zeneca |
CIs: Dr Rob Jones/ Dr Sacha Howells |
|
FURVA
(closed) |
Vandetanib plus fulvestrant versus placebo plus fulvestrant after relapse or progression on an aromatase inhibitor in metastatic ER positive breast cancer (FURVA): A randomised, double-blind, placebo-controlled, phase II triaLhttps://www.annalsofoncology.org/article/S0923- 7534(20)42330-0/fulltext
Funder: CRUK and Astra Zeneca |
CIs: Dr Rob Jones/ Dr Mark Beresford |
|
EAGLE
(closed) |
EAGLE: Improving the wellbeing of men by addressing the late effects of radical treatment for prostate cancer |
Professors John Staffurth/Ann- Marie Nelson |
|
ROCS
(closed) |
Palliative radiotherapy in addition to self-expanding metal stent for improving outcomes of dysphagia and survival in advanced oesophageal cancer |
Professor Anthony Byrne/Dr D Adamson |
|
ALERT_B
(closed) |
A screening tool for the detection of gastroenterological late effects after radiotherapy for prostate cancer. bmjopen.bmj.com/content/bmjopen/6/10/e011773.full.pdf |
Authors Professors John Staffurth/Ann- Marie Nelson |
|
COBRA |
Patient Reported Core Outcomes in Brain Tumour Trials
Funder: The Brain Tumour Charity |
Professor Anthony Byrne |
[Studies led by Velindre Chief Investigators sponsored by other organizations not included]
Appendix 4. Current Clinical Trial Portfolio VUNHST
4A: Portfolio of Clinical Trials led (by Velindre Chief Investigators) and Sponsored by Velindre (open, in set-up and in follow-up).
4B: Percentage of new Velindre referrals recruited to Clinical Trials 2015-2020
4C: Portfolio of Clinical Trials Hosted by Velindre – by type and phase
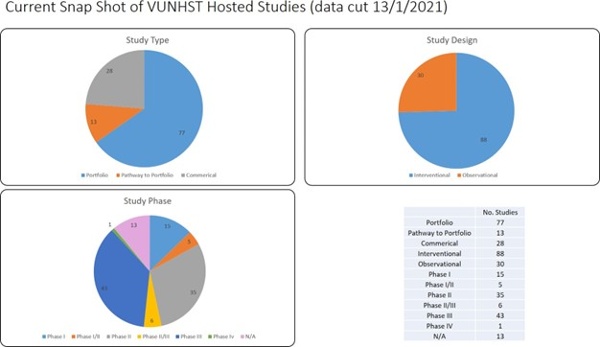
4D: Portfolio of Clinical Trials Hosted by Velindre – by tumour site (with Velindre Principal Investigators)